Today is World Water Monitoring Day! Participate by ordering a test kit and submitting sample data through December of this year. Also, check out the ocean of other water citizen science projects on SciStarter.
Here at SciStarter, we spend a lot of time supporting citizen science, but we also happen to be citizen scientists ourselves. In the spirit of World Water Monitoring Day, I trekked to the Charles River in Boston to grab a water sample. Barring all potential parking and trespassing violations, it was a success! Still, you might wonder, why does this sample matter? Why care about water?
I’m glad you asked. But before I dive deeper (pun intended), here are some facts to consider. An adult human is made of ~60% water. About 70% of Earth is covered by water. We need water for our metabolic processes internally and for our day-to-day tasks externally. Water is there when you shower, brush your teeth, or guzzle down a drink after a run. Water is also essential for the productivity of farms, which, in turn, provide us food. You get the picture: we need water. Likewise, so do other animals and plants, especially those that live in or near aquatic environments.
Consequently, the sample data collected and submitted by millions of people on World Water Monitoring Day not only benefit us human beings. It also helps scientists better understand a multitude of aquatic environments around the globe.
Participating couldn’t be easier. World Water Monitoring Challenge, an education and outreach program, provides kits that you can purchase and use to sample the water in your area. Here are the main concepts behind what you can test and why it’s important to do so.
Turbidity, the measure of relative water clarity. This is important when producing drinking water for human consumption and for many manufacturing uses. Turbid water may be the result of soil erosion, urban runoff, algal blooms, and bottom sediment disturbances caused by boat traffic and bottom-feeding fish. (You can even make your own secchi disk to measure turbidity.)
pH, a measurement of the acidic or basic quality of water. Most aquatic animals are adapted to a specific range of pH level and could die, stop reproducing, or move away if the pH of the water varies beyond their range. Low pH levels can also allow toxic compounds to be exposed to aquatic plants and animals. pH can be affected by atmospheric deposition (acid rain), wastewater discharge, drainage from mines, or the type of rock in the surrounding area.
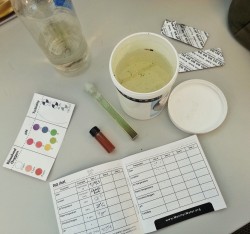
Dissolved oxygen levels. Natural water with consistently high dissolved oxygen levels is most likely to sustain stable and healthy environments. Changes to aquatic environments can affect the availability of oxygen in the water. High levels of bacteria or large amounts of rotting plants can cause the oxygen saturation to decrease, which affects the ability of plants and animals to survive in and around it.
Water temperature. If temperatures are outside an organism’s normal range, the organism could become stressed or potentially die. Temperature also affects the rate of photosynthesis in aquatic plants as well as their sensitivity to toxic wastes, parasites, and disease. Furthermore, water temperature can affect the amount of oxygen water can hold (cold water holds more oxygen than warm water).
This project is ideal for anyone who lives near a water source, educators who want ideas to teach students about water chemistry, or citizen scientists hoping to contribute to an increasingly important field of research.
It’s the perfect project to illustrate that when it comes to citizen science, you can dive right in.
References:
“How Much Water is There On, In, and Above Earth?” USGS. Web. 9/18/13
“Importance of Turbidity.” Environmental Protection Agency. 9/18/13
“The Water in You.” USGS. Web. 9/18/13
World Water Monitoring Challenge booklet
“World Water Monitoring Day.” Wikipedia. Wikimedia Foundation, Inc. Web. 9/18/13
Images: Lily Bui
Lily Bui is the executive editor of SciStarter. She holds dual degrees in International Studies and Spanish from the University of California Irvine. She has worked on Capitol Hill in Washington, D.C.; served in AmeriCorps in Montgomery County, Maryland; worked for a New York Times bestselling ghostwriter; and performed across the U.S. as a touring musician. She currently works in public media at WGBH-TV and the Public Radio Exchange (PRX) in Boston, MA. In her spare time, she thinks of cheesy science puns. Follow @dangerbui.

